Ortho-fluoro aniline and pH reactions
Moderators: Xen, expert, ChenBeier
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Ortho-fluoro aniline and pH reactions
Consider ortho-fluoro aniline (o-FC6H4NH2).
a.Give an electron-dot formula for this compound and indicate with an arrow which atoms can accept a proton. Circle the atom that actually does accept a proton from water.
b.Determine whether o-FC6H4NH2 is a stronger or weaker base than aniline itself (C6H5NH2) and explain why this is the case.
c.Calculate the pH of a 0.1 M solution of o-FC6H4NH2.
d.Draw the structural formula of the conjugate acid of o-FC6H4NH2.
e.Calculate the pH of a 0.010 M solution of the conjugate acid of o-FC6H4NH2.
What are answers to all these questions? How to answer all these questions? Let me answer all these questions one by one.
a.Give an electron-dot formula for this compound and indicate with an arrow which atoms can accept a proton. Circle the atom that actually does accept a proton from water.
b.Determine whether o-FC6H4NH2 is a stronger or weaker base than aniline itself (C6H5NH2) and explain why this is the case.
c.Calculate the pH of a 0.1 M solution of o-FC6H4NH2.
d.Draw the structural formula of the conjugate acid of o-FC6H4NH2.
e.Calculate the pH of a 0.010 M solution of the conjugate acid of o-FC6H4NH2.
What are answers to all these questions? How to answer all these questions? Let me answer all these questions one by one.
Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Re: Ortho-fluoro aniline and pH reactions
Answer to a.
Chemical structure of ortho-fluoro aniline is as follows:
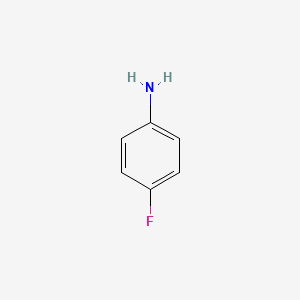 Nitrogen atom can accept proton from water.
Nitrogen atom can accept proton from water.
Answer to b.
o-FC6H4NH2 is stronger base than C6H5NH2 because F can act as base (electron pairs donor)
Chemical structure of ortho-fluoro aniline is as follows:
 Nitrogen atom can accept proton from water.
Nitrogen atom can accept proton from water.Answer to b.
o-FC6H4NH2 is stronger base than C6H5NH2 because F can act as base (electron pairs donor)
Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- ChenBeier
- Distinguished Member

- Posts: 1569
- Joined: Wed Sep 27, 2017 7:25 am
- Location: Berlin, Germany
Re: Ortho-fluoro aniline and pH reactions
Both are wrong . What you draw is para fluor aniline not the ortho one.
And anilin pka 4,6 is stronger base as the fluoraniline pka 3,2. Reason strong - I effect of flourine, M - effect is miner.
And anilin pka 4,6 is stronger base as the fluoraniline pka 3,2. Reason strong - I effect of flourine, M - effect is miner.
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Re: Ortho-fluoro aniline and pH reactions
Are Yoy talking about mesomeric effect and inductive effect of Fluorine?
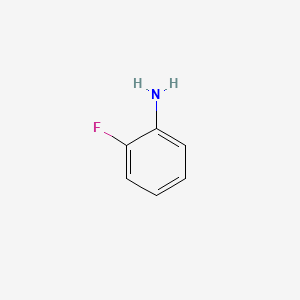 Is this correct chemical structure drawing for ortho-fluoro aniline?
Is this correct chemical structure drawing for ortho-fluoro aniline?
 Is this correct chemical structure drawing for ortho-fluoro aniline?
Is this correct chemical structure drawing for ortho-fluoro aniline?Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- ChenBeier
- Distinguished Member

- Posts: 1569
- Joined: Wed Sep 27, 2017 7:25 am
- Location: Berlin, Germany
Re: Ortho-fluoro aniline and pH reactions
Now it is correct. Yes i talk about inductive and mesomeric effect.
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Re: Ortho-fluoro aniline and pH reactions
Fluorine atom will accept proton from water in ortho-fluoro aniline.
You said pKa of the o-fluoro aniline is 3.2, so its pH is 3.2 as well.
Now, how to answer part d and e?
You said pKa of the o-fluoro aniline is 3.2, so its pH is 3.2 as well.
Now, how to answer part d and e?
Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- ChenBeier
- Distinguished Member

- Posts: 1569
- Joined: Wed Sep 27, 2017 7:25 am
- Location: Berlin, Germany
Re: Ortho-fluoro aniline and pH reactions
Wrong. The only atom which accept proton will be nitrogen to form the ammonium.
The calculation is similar the first exercise with the trimethylamin.
The calculation is similar the first exercise with the trimethylamin.
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Re: Ortho-fluoro aniline and pH reactions
Let me answer part e.
Conjugate acid of o-FC6H4NH2 is o-FC6H4NH3
Reaction equation is FC6H4NH2 + H2O → FC6H4NH3⁺ + OH⁻
So, 1.6E-11=x*x/0.01M → x= 4.0e-7, Hence pOH=-log(4.0e-7)= 6.4. so, pH=14.0-6.4=7.6 Is this computaion correct?
Conjugate acid of o-FC6H4NH2 is o-FC6H4NH3
Reaction equation is FC6H4NH2 + H2O → FC6H4NH3⁺ + OH⁻
So, 1.6E-11=x*x/0.01M → x= 4.0e-7, Hence pOH=-log(4.0e-7)= 6.4. so, pH=14.0-6.4=7.6 Is this computaion correct?
Last edited by Dhamnekar Winod on Mon Mar 22, 2021 4:53 am, edited 1 time in total.
Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- ChenBeier
- Distinguished Member

- Posts: 1569
- Joined: Wed Sep 27, 2017 7:25 am
- Location: Berlin, Germany
Re: Ortho-fluoro aniline and pH reactions
Where does the 0,00625 coming from. pH should be alkaline because its a amin
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Re: Ortho-fluoro aniline and pH reactions
I edited the answer. Please check it out.
Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- ChenBeier
- Distinguished Member

- Posts: 1569
- Joined: Wed Sep 27, 2017 7:25 am
- Location: Berlin, Germany
Re: Ortho-fluoro aniline and pH reactions
I dont know what you doing, where this numbers come from. pKa is 3.2 already
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Re: Ortho-fluoro aniline and pH reactions
If pKa is 3.204, then ka= 0.000625, this means 0.000625* 0.01= x * x , → x= 0.0025 Now is this hydroxide ion concentration? If it is so, then poH=-log(0.0025)=2.602, this means pH= 14.000 -2.602=11.398 Is this computation correct?
Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- ChenBeier
- Distinguished Member

- Posts: 1569
- Joined: Wed Sep 27, 2017 7:25 am
- Location: Berlin, Germany
Re: Ortho-fluoro aniline and pH reactions
Looks better now
- Dhamnekar Winod
- Distinguished Member

- Posts: 257
- Joined: Sat Nov 21, 2020 10:14 am
- Location: Mumbai[Bombay] and Mumbai Suburb,, Maharashtra State,India
Re: Ortho-fluoro aniline and pH reactions
How to use pH solver available on webqc.org to answer this question?
Any science consists of the following process. 1) See 2)Hear 3)Smell,if needed 4)Taste, if needed 5)Think 6)Understand 7)Inference 8)Take decision [Believe or disbelieve, useful or useless, true or false, cause or effect, any other criteria]
- ChenBeier
- Distinguished Member

- Posts: 1569
- Joined: Wed Sep 27, 2017 7:25 am
- Location: Berlin, Germany
Re: Ortho-fluoro aniline and pH reactions
The given pKs is 3.2 for FC6H4NH3+ , for the solver has to be used the corresponding pKb value of 10.8.
pH about 8.1
pH about 8.1
