Page 1 of 1
Org structures help
Posted: Fri Apr 02, 2021 4:59 am
by nike_victoria
Help me please and thank you. Redraw acetaminophen on your paper and draw the non-bonding electron pairs if any. Encircle the site with the most delocalized electrons. Then, identify the hybridization of all the carbons in the structure.
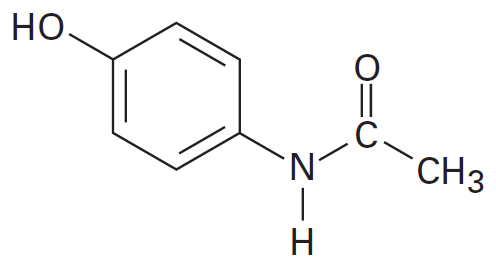
- 952ACA45-E87B-4EC4-9747-56C93A83B2B2.png (8.17 KiB) Viewed 1217 times
Re: Org structures help
Posted: Fri Apr 02, 2021 6:28 am
by ChenBeier
Each oxygen has two and nitrogen one electron pair.
4 Single bond is sp3, one double bond and 2 single bond is sp2 and 1 tribond and 1 Single bond or two double bond is sp hybrid.